Lately scientists are finding atoms that may represent the shores of this island.Įlement 114, for instance, decays somewhat less quickly than some calculations had predicted that an atom jammed with that many protons must. Thanks to special configurations inside their nuclei that grant unusual stability, superheavy elements inhabiting this region may not be just ephemeral creations of the laboratory but could actually stick around. If so, they would form a long-sought region on the periodic table called the island of stability. Some of our envisioned elements may linger for minutes, maybe even years without decaying. But scientists suspect that certain yet to be discovered superheavy elements and isotopes (versions of the same element with different numbers of neutrons) might break this teasing pattern of fleeting, tantalizing existence. The few atoms we make exist only for brief moments before collapsing or transforming under the strain of too many positively charged protons repelling one another. 
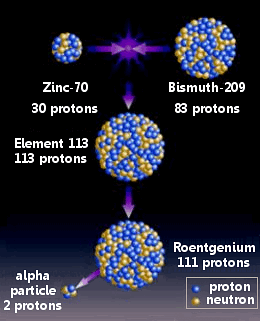
Each new species we find is exciting because it represents an unknown material, a form of matter humans have never encountered before. One of us (Düllmann) has been conducting some of the first chemistry experiments on several of the so-called superheavy elements, and Block has been working on the first direct mass measurements and other investigations into some of them. At the same time that oganesson became official, researchers also added elements containing 113, 115 and 117 protons per atom to the periodic table. Each one of the fundamental bits of nature on the table is defined by the number of protons packed in its atomic nucleus. It was named for Yuri Oganessian of the Russian-based institute, who is a pioneer of this research.īut how many more elements are out there? In just the past decade scientists have been pushing the periodic table further and further, adding new atoms that are heavier than ever before. In 2015, after more than a decade of vetting and rechecking, element 118 officially joined the periodic table of the elements, the world's master list of matter. After 1,080 hours of collisions, the investigators had created three atoms of this new superheavy substance.īut by carefully accounting for all of the radiation and smaller atoms that the reactions produced, the scientists at the Joint Institute for Nuclear Research in Russia could be fairly sure that they had, for a brief sliver of a moment, created the element. Scientists announced the discovery of oganesson in 2006, when a Russian-American team used a particle accelerator in Dubna, Russia, to fire millions of trillions of calcium ions at a target of heavy atoms. In contrast, hydrogen-the most abundant element in the universe, something you can find in your body, Earth's oceans and even the atmosphere of Jupiter-has only one. Each atom of the stuff packs a whopping 118 protons into its dense center. The heaviest element that humans have ever found is called oganesson. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
